
By Chris Taylor
About the Author
Chris Taylor is a Senior Engineer at Aeson Power, specializing in sodium-ion cell design and performance optimization. His research focuses on high-rate discharge behavior, thermal stability at extreme high temperatures, low-temperature adaptability, and real-world operational performance of sodium-ion batteries.
At its core, every battery is a materials problem. The electrodes, electrolyte, and interfacial structures responsible for storing and releasing energy are all directly affected by temperature: at low temperatures, the ion desolvation process slows and ionic transport resistance increases; at high temperatures, the cathode's microscopic crystal structure may become destabilized. For any starting battery deployed in extreme climates, the fundamental question is: which chemistry fails least at the materials level, and across the widest possible temperature range?
This article explores sodium-ion batteries (SIBs) and their application in professional 12V and 24V starting battery products. For years, we have been working to solve and optimize a persistent problem: why do mainstream energy storage solutions fail exactly when they are needed most? Whether it is lead-acid batteries unable to start vehicles at -20°C, or lithium iron phosphate (LFP) batteries with limited cranking output at 50°C, the narrow temperature operating window has long been the “Achilles’ heel” of electrified applications. Sodium-ion battery technology is fundamentally redefining this boundary.
The Core of Wide-Temperature Performance: Kinetics and Stability
The key barrier to reliable starting in extreme temperatures lies in the battery’s internal resistance — specifically, its ability to maintain high stability when temperatures fluctuate dramatically. Sodium-ion chemistry possesses a natural advantage here, enabling it to sustain excellent stability across both high and low temperature extremes.
At the low-temperature end, the critical factor is desolvation energy. Before ions can enter the electrode, they must shed the solvation shell surrounding them. Compared to Li⁺, Na⁺ has a smaller Stokes diameter, which means sodium ions naturally exhibit higher ionic mobility in electrolytes — a structural kinetic advantage confirmed by Bai et al. in a 2024 review published in Advanced Energy Materials. A 2025 review in EES Batteries (Royal Society of Chemistry) further confirmed that SIBs demonstrate superior capacity retention and cycling stability below -20°C compared to LFP, attributed to sodium’s lower charge density and faster interfacial kinetics in sub-zero conditions.
In severe cold, extremely low temperatures cause lithium-ion mobility to drop sharply, making intercalation into electrodes difficult and causing internal resistance to spike. Under such conditions during charging, lithium ions may fail to intercalate into the electrode interior and instead deposit on the anode surface, increasing lithium plating and creating serious safety risks.
Sodium ions, by contrast, remain highly active in low-temperature environments, delivering stable current output even at -40°C with very high capacity utilization. Optimized SIB cells maintain capacity retention above 85% across a temperature range of -30°C to 60°C — a performance envelope that closely matches the real-world demands of extreme-climate starting applications.
At the high-temperature end, sodium-ion materials demonstrate superior thermal resilience. The microscopic crystal structure of our cathode materials is more robust, and significantly less prone to accelerated reactions when heated. This intrinsic stability means the battery not only survives high-temperature environments, but continues to operate reliably without the rapid aging or adverse safety risks seen in other chemistries.
From an engineering perspective, this enables what we call "Engineering Subtraction": because this chemistry naturally accommodates temperatures from -40°C to 80°C, it allows us to significantly simplify the Battery Management System (BMS) and eliminate bulky, energy-consuming active heating and cooling circuits. This powerful wide-temperature self-adaptability greatly reduces structural complexity and delivers meaningful cost advantages.
According to IRENA (International Renewable Energy Agency)’s sodium-ion battery report released in November 2025, sodium carbonate — the key raw material precursor — was priced between just $100 and $500 per tonne from 2020 to 2024, while lithium carbonate reached $6,000 to $83,000 per tonne over the same period. Furthermore, sodium is approximately 1,000 times more abundant than lithium in the earth’s crust. At the cell level, SIB designs can substitute lower-cost aluminum for the copper anode current collector used in traditional lithium cells, further reducing material input costs. IRENA projects that with scale-up, SIB cell costs could fall to $40/kWh. Combined with a simplified BMS architecture, this makes SIBs a highly cost-effective long-term option for starting battery applications.
Head-to-Head Comparison: SIB vs. LIB vs. Lead-Acid
These findings are consistent with peer-reviewed literature: both the 2025 review in EES Batteries and the 2024 study in Nanomaterials confirm that SIBs exhibit stronger discharge capability and cycling stability at sub-zero temperatures compared to LFP. The table below presents Aeson Power’s laboratory performance benchmark data:
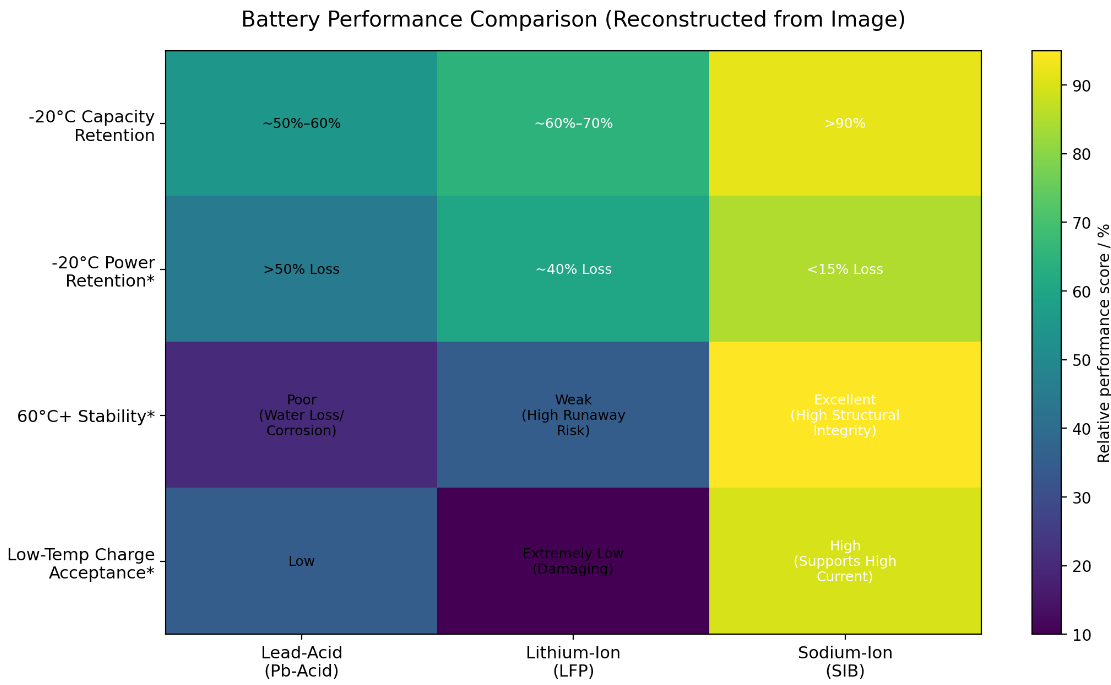
Data Note: All data shown is based on laboratory testing conducted by Aeson Power under controlled conditions. Actual performance may vary depending on cell design, system configuration, state of charge, and application environment.
Aeson Power in the Field: From Arctic Cold to Tropical Heat
Aeson Power is translating these advantages into real-world product capability in the professional starting battery market.
In high-latitude cold regions, Aeson Power sodium-ion starting batteries deliver a reliable low-temperature cranking solution. Unlike lead-acid batteries that struggle in sub-zero conditions due to rising internal resistance, our 12V and 24V products can still deliver sustained, stable cranking current at -40°C, and maintain more than 90% capacity at -20°C. This ensures first-crank success and reliable backup power capacity for both passenger cars and heavy commercial vehicles — without any external heating.
At the same time, Aeson Power has achieved significant success in Southeast Asian markets, where high temperatures are the primary battery killer. In tropical climates, traditional starting batteries commonly fail within a year due to accelerated corrosion and electrolyte loss. Sodium-ion batteries, with their superior structural stability, deliver a cycle life more than 5 times that of comparable lead-acid products at elevated temperatures, making them ideally suited for this region. Aeson Power batteries are now experiencing rapid growth in multiple Southeast Asian countries, demonstrating their ability to perform reliably under consistently high ambient temperatures.
Conclusion: Ending Temperature Anxiety
For decades, engineers compensated for battery limitations by adding heaters and coolers to keep cells within their “comfort zones.” Sodium-ion technology allows us to stop this futile compensation and start genuinely innovating.
With superior interfacial kinetics and structural thermodynamics, Aeson Power’s sodium-ion products are proving that the powertrains of the future — from frozen tundra to tropical zones — need no longer be constrained by extreme environments. The data is clear: where lead-acid and LFP fall short, SIBs deliver stable output — exceeding 90% capacity retention at -20°C, operational capability at -40°C, and structural integrity above 60°C. Sodium-ion batteries are becoming the new standard for resilience in demanding applications.
To learn more about Aeson Power’s sodium-ion batteries, please visit www.aesonpower.com.



















